¡Búscanos en las redes sociales!

Advocacy for Quality:
Patient Demand for Evidence-Based Substitution of Biosimilars
Since 2008, Rituximab has been the primary biologic agent used in Panama’s Rheumatology Service.
A 2019 Executive Order initially protected patients on the innovator product from mandatory substitution, but it was repealed in 2022.
The crisis emerged when CSS mandated replacement with biosimilars starting in 2019. Though Panama approved two rituximab biosimilars—one low-performing and one high-quality—CSS chose the inferior option to save just $20 per dose.
This low-quality biosimilar immediately caused adverse effects for patients.
How we did it
- Formal meetings with RMD Service in 2019 – no positive outcomes.
- Submitted a protection of constitutional rights at the Supreme Court in 2019
- High-Visibility Media: FUNARP protested in the CSS hospital, appeared on the TV, and on local newspapers, rejecting the arbitrary substitution.
- Formal Communication: Met with and sent letters to the ombudsman in 2003.
- Formal meetings with the Head of the Rheumatology Service, citing legal requirements – positive outcome
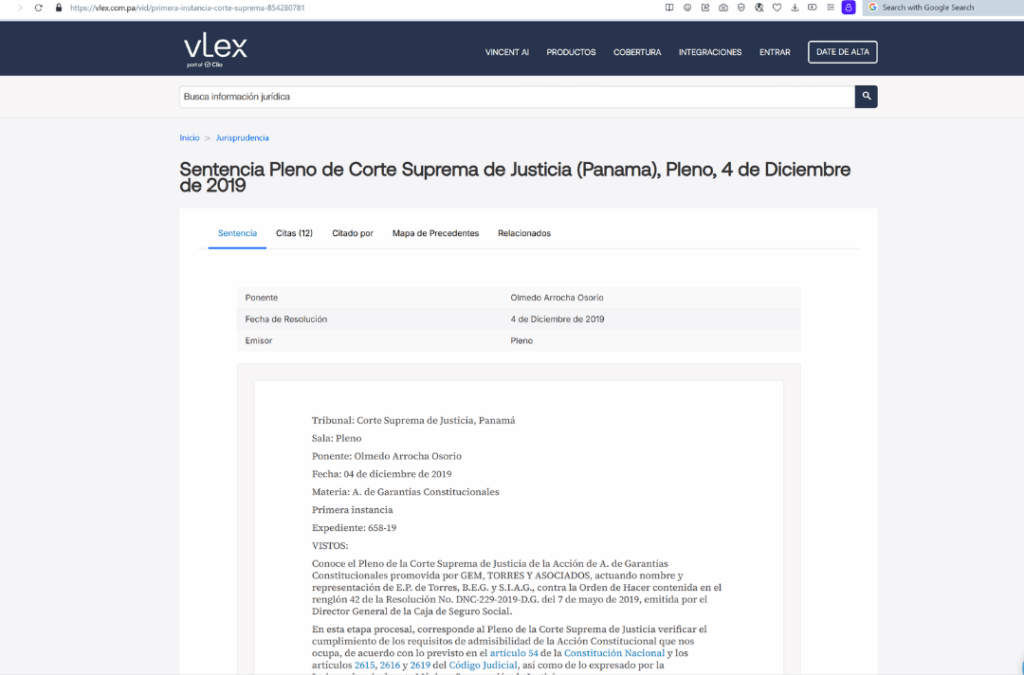
2019 -Action of unconstitutionality
FUNARP lawyers filed an action of unconstitutionality with the Panamanian Supreme Court of Justice
Original publication – click here
English translation:
Panama’s Supreme Court reviewed a constitutional protection action against the CSS Resolution ordering substitution of the innovator biologic with a biosimilar of inferior quality through public bidding.
Rheumatoid arthritis patients argued that Executive Decree 105 of 2017 prohibits interchangeability of biologics due to immunogenicity risks.
They contend the switch would endanger their health after years of successful treatment with the original medication.
This action was not admitted due to technicalities.
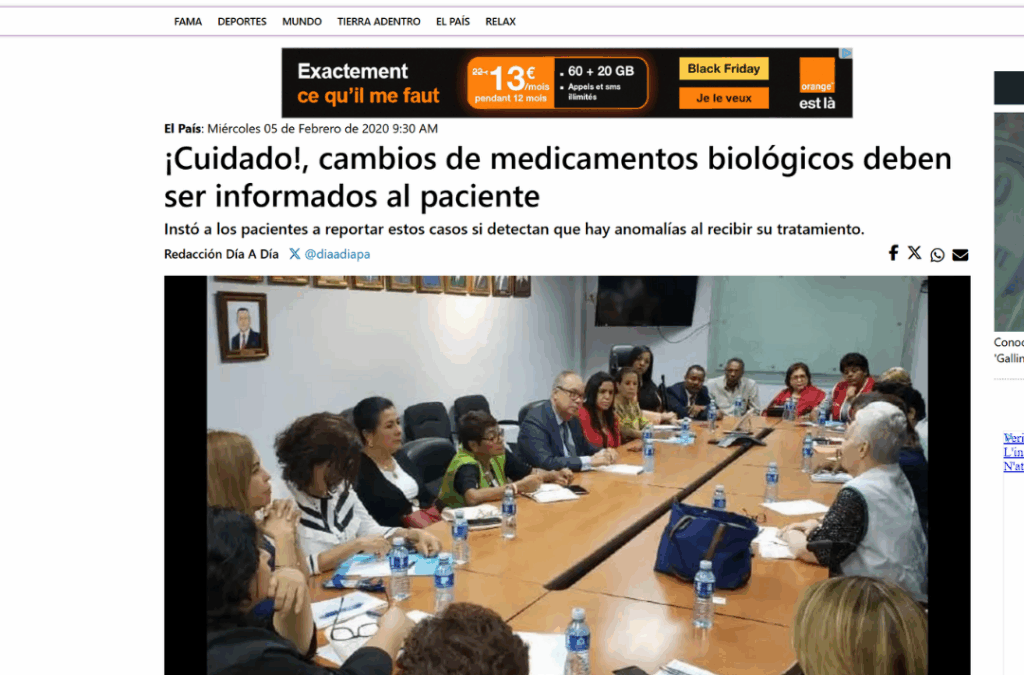
2020 -Newspaper article
Caution! Changes of biologic medications must be informed to the patient
Original publication – click here
English translation:
The president of FUNARP explained that in the case of biologic medications, the patient must be vigilant regarding any medication substitution or automatic substitution attempt that could alter their treatment.
“It is clearly prohibited that at the point of dispensation, that is, at the pharmacy, biologic medications that the patient is using be substituted with biosimilars. Especially in critical and chronic diseases,” emphasized Enma Pinzón, representative of the Rheumatoid Arthritis Foundation of Panama (Funarp).
According to Pinzón, this is a matter of utmost importance, which is why Funarp maintains a vigilant attitude regarding patients’ rights to ensure the quality, safety, efficacy, and continuity of therapies available to Panamanians.
For Funarp, patients have the right and obligation to know in detail each medication they receive and why the doctor prescribes it. This way, in addition to having better knowledge of their disease management, they can actively participate in pharmacovigilance, meaning they can report any symptom or adverse effect and can alert their doctor immediately.
She explained that in the case of biologic medications, the patient must be vigilant regarding any medication substitution or automatic substitution attempt that could alter their treatment.
That being the case, it is concerning that the Social Security Fund (CSS), despite citing the decree as the legal foundation of the bidding process, does not comply with it. Subsequently, it has promised to respect the therapy for existing patients; however, more than seven months have passed and inventories in some cases are depleted and others are about to be depleted at the institution, seriously jeopardizing the continuity of therapy and with it the health of patients.
According to Funarp, patients must ensure that what is stipulated in the decree is fulfilled: for the prescription of non-substitutable medications, the prescription must contain the generic name of the product, the commercial name in parentheses, and the phrase “Non-Substitutable.”
Pinzón clarified that “it is important for patients to know that biosimilar medications that enter Panama, according to the law, must have approvals from high-standard countries; however, they are for use with new patients.”
She urged patients to report these cases if they detect any anomalies when receiving their treatment.
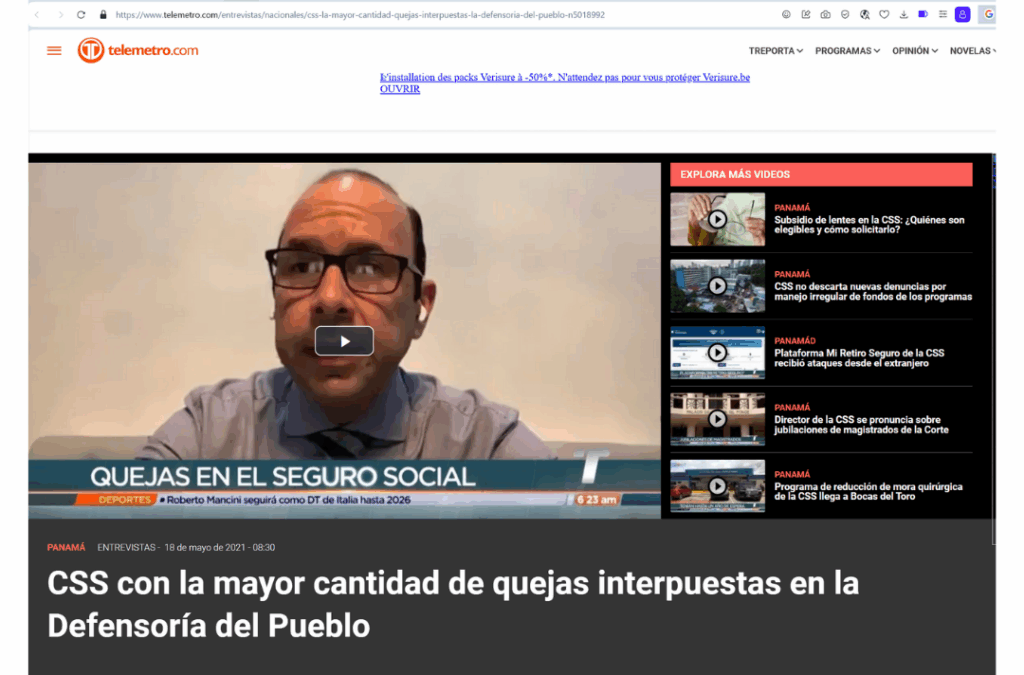
2021 -CSS with the highest number of complaints filed with the Ombudsman's Office
FUNARP lawyers filed an action of unconstitutionality with the Panamanian Supreme Court of Justic
Original publication – click here
English translation:
CSS with the highest number of complaints filed with the Ombudsman’s Office
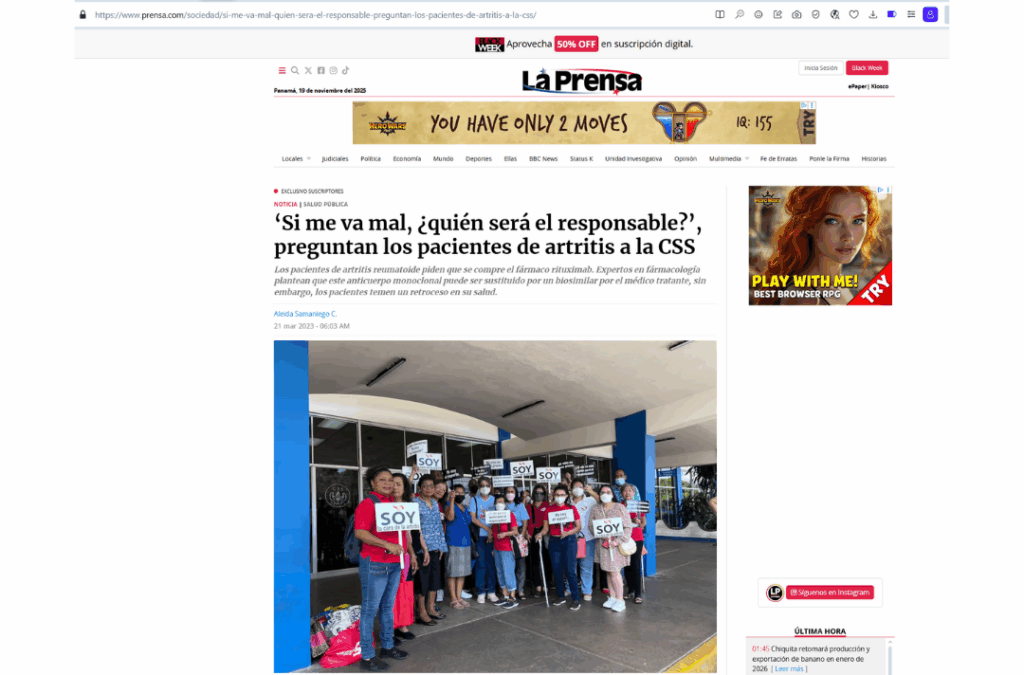
2023 -Protest at the Social Security Fund Hospital
If things go wrong for me, who will be held responsible?” ask arthritis patients to the CSS
Original publication – click here
English translation:
Panama’s Supreme Court reviewed a constitutional protection action against the CSS Resolution ordering substitution of the innovator biologic with a biosimilar of inferior quality through public bidding.
Rheumatoid arthritis patients argued that Executive Decree 105 of 2017 prohibits interchangeability of biologics due to immunogenicity risks.
They contend the switch would endanger their health after years of successful treatment with the original medication.
This action was not admitted due to technicalities.
2023 - TV Appereances
FUNARP Explains the Rejection by Some Patients to the Change of Medication for Treating Arthritis
Original publication – click here
English translation:
Emma Pinzón, president of the Rheumatoid Arthritis Foundation of Panama (FUNARP), explains the rejection that some patients maintain regarding the change of medications to treat arthritis.
2023-2025 - Meetings with the RMD Service of the Social Security Fund Hospital Chief and doctors
- Formal meetings with the Head of the Rheumatology Service, citing legal requirements and expert physician concerns regarding multiple cases of therapeutic failure and citing legal obligations to protect patients.
OUTCOME
The Head of the Department sent the letter to the CSS administration to stop the purchase of the low-quality biosimilar and to request the purchase of the better alternatives.
NEXT STEPS
- Policy Compliance Monitoring to ensure they act immediately upon the collegial clinical consensus of the Rheumatology Service.
- Pharmacovigilance Awareness Campaign for patients with the slogan “I am the report”.
- To meet with police makers to create a law to protect patients.
